Can’t-Miss Takeaways Of Info About How To Draw Covalent Bonds

Determine the number of electrons necessary to satisfy the octet (or duet) rule with no electron sharing.
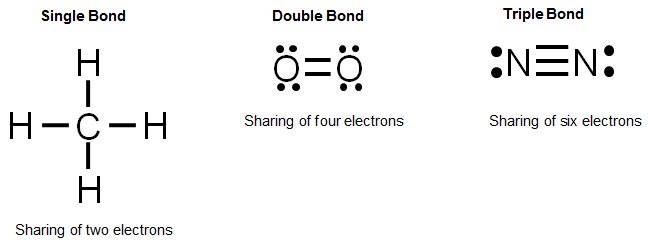
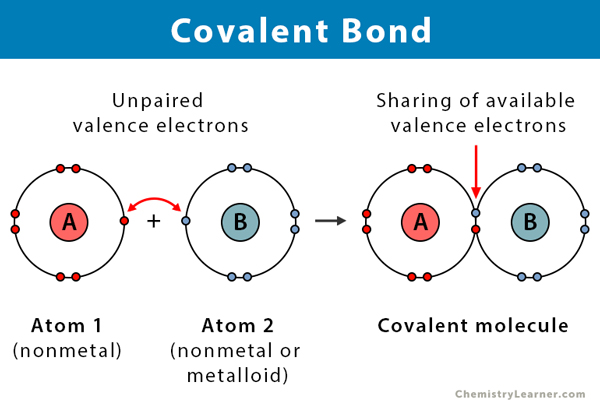
How to draw covalent bonds. It is often easiest to draw circles at 90° or 180° to each other. Draw a lewis dot structure for the valence. A covalent bond is a chemical bond that comes from the sharing of one or more electron pairs between two atoms.
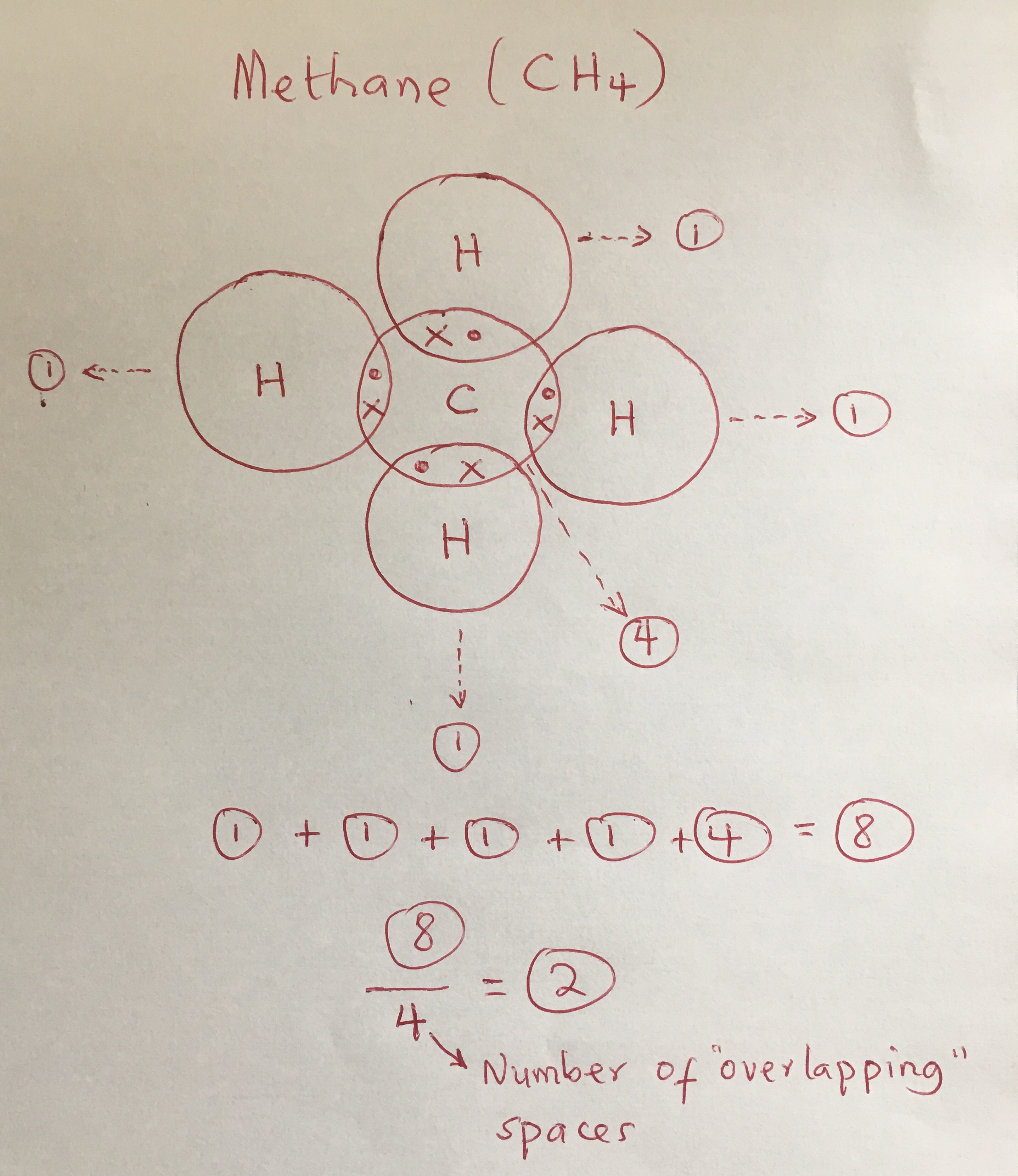
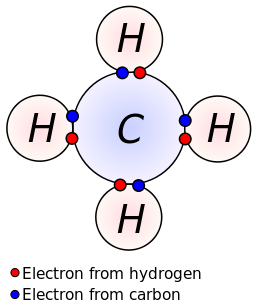
Determine the number of valence electrons available. Sharing of electrons between atoms of the same kind e.g. Practice covalent compounds (molecular compounds):• h₂o water (dihydrogen monoxide)• o₂ oxygen• co₂ carbon dioxide• hf hydrogen fluoride• ch₄ methane• p.
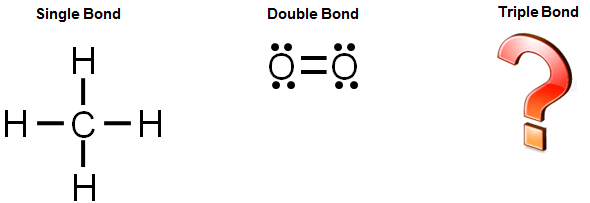
There are different kinds of covalent bonds: A single covalent bond is when two atoms share a single pair of electrons. It is often easiest to draw circles at 90° or 180° to each other.
There are three shared spaces between the circles, so add a dot and. Contents:0:08 introduction 0:39 h21:25 hcl2:23 cl2. In covalent bonds, atoms share electrons.
This is because of the unequal sharing of electrons (due to the difference in electronegativity) between oxygen and hydrogen. Covalent bonding can be achieved in two ways: There are three shared spaces between the circles, so add a dot and.
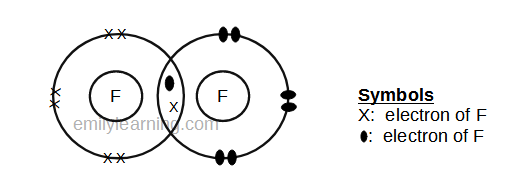
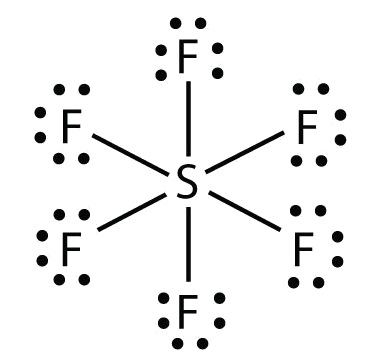
In these diagrams, we use circles to represent the electron shells. Formation of covalent bonds we draw dot and cross diagrams with overlapping valence electron shells, to illustrate how covalent bonds are formed. Nitrogen is in group 5 so it forms three covalent bonds.
The diagrams below illustrate the. Sharing of electrons between atoms of different kind. To learn about covalent bonds, let’s start by looking at the structure of a molecule of hydrogen.
Water is a polar covalent molecule. Use one line for each pair of electrons that is shared.